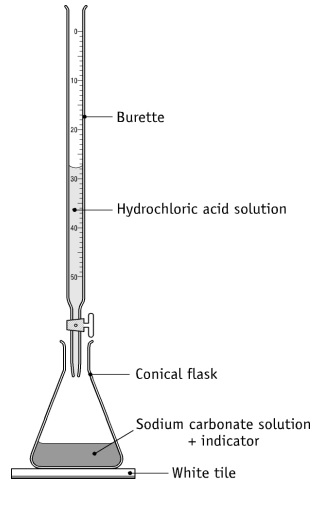
D ) (DJU FUR) 1 cc of 0.1 N HCl is added to 99 cc solution of NaCl. The pH of the resulting solution will be (A) 7 (B) 3 (D) 1 M (C) 4 M

1 normal solution of hcl | 1 N solution of hcl | 1 N hcl | hydrochloric acid solution preparation - YouTube
One litre solution of N/2 HCl is heated in a beaker. It was observed that when the volume of solution is reduced to 600 mL , 3.25 g of HCl is lost.
62. On adding which of the following,the pH of 20 ml of 0.1N HCl will not alter? a)20 ml of distilled water b)1ml of .1N NaOH c)500 ml 0f HCl of Ph=1

20 ml of 1 N HCI, 10 ml of H,80, and 30 ml of HNO3 are mixed together and volume made to one litre. The normality of He in the resulting

How To Prepare 1N And 0.5N Hydrochloric Acid(HCl)||Preparation Of Normal Solution||#Chemistrycubicle - YouTube